The effect ofpressure on the enthalpy of natural gases is dependent upon the pressure-volume-temperature relationships. 
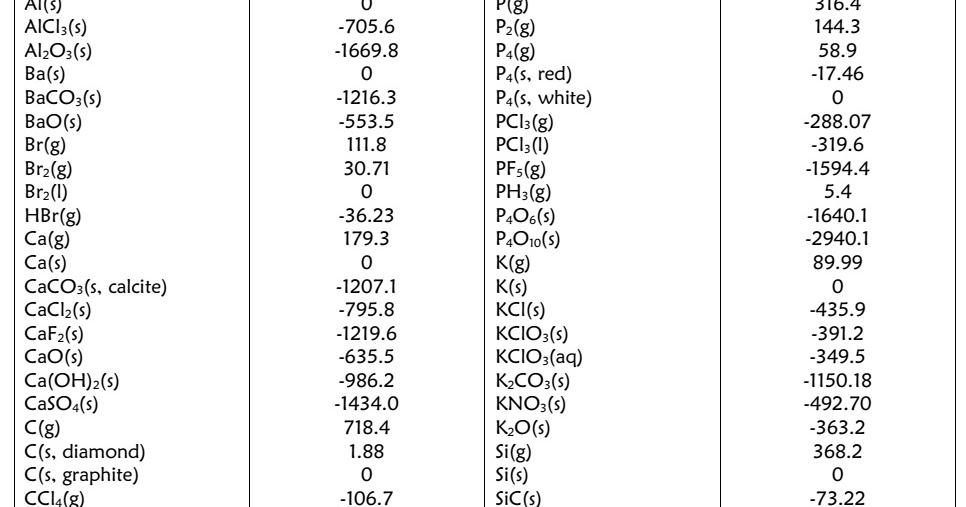
Fromthis it is clear that the specific heat of natural gases is a function only ofthe gas gravity and the temperature at atmospheric pressure. The best available datafor natural and petroleum refinery gases indicates the relationship shown. The effect of temperature upon the enthalpy at consistent pressure is expressedas the ?heat capacity? or ‘specific heat’ of the gas. A careful study ofthe known properties of natural gas indicates that this is possible. For this reason it would beextremely convenient of a reasonably satisfactory enthalpy-entropy diagramcould be prepared as a function of the gravity of the gas. The enthalpy-entropy diagram for natural gases is to the gas engineer what thesteam diagram is to the steam-power engineer. The power requiredfor the compression of the gas may be readily determined by converting theincrease in enthalpy into the desired units. When compression or expanding a gas by which means of a compressor or engine inwhich no heat is added to or subtracted from the gas, but only work done, thechanges in the properties of the gas may be determined along a vertical line ofconstant entropy between the entering and exit pressures. Information theory finds applications in machine learning models, including Decision Trees. Events with higher uncertainty have higher entropy. In information theory, a random variable’s entropy reflects the average uncertainty level in its possible outcomes. Under such conditions the changes in theproperty of the fluid as it flows through a throttling valve, choke, or anyother similar arrangement, may be read directly from the enthalpy-entropydiagram by following a horizontal line between the known pressures. Entropy measures the amount of surprise and data present in a variable. the entropy of a pure substance at 298 K and 1 atm pressure). Standard molar entropies are listed for a reference temperature (like 298 K) and 1 atm pressure (i.e. Well that’s exactly how and why decision trees use entropy and information gain to determine which feature to split their nodes on to get closer to predicting the target variable with each split and also to determine when to stop splitting the tree ( in addition to hyper-parameters like max depth of course). In cases where there is no significant change in potential energy or in kineticenergy (velocity), it follows that the increase in enthalpy is equal to thetotal energy supplied to the fluid. Values of the standard molar entropies of various substances at 298 K (25☌) are given in the table. The entropy of a substance has an absolute value of 0 entropy at 0 K. In dealing with the flow of fluids, the sum of the increasein the enthalpy plus the increase in kinetic energy plus the increase inpotential energy of the fluid in flow, is equal to the sum of the heat and workadded to the fluid while flowing between the entrance and exit of the flowsystem. The Mollier diagram in which the enthalpy (heat content) is plotted against theentropy with lines of constant temperature, pressure, and in some cases volume, has been found most convenient when dealing with the compression, expansion, and flow of fluids. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
